|
The IMF is maintained out of equilibrium in respiring cells by transmembrane (TM) complexes that actively pump ions outward across the inner membrane. Where ΔΨ = Ψ int−Ψ ext is the difference in membrane potential Ψ, built up by the ensemble of charges separated by the membrane, Δ p I = p I i n t - p I e x t = log e x t i n t is the difference in the specific ion ( I) potential, is the ion concentration, R is the gas constant, F is the Faraday constant, and T is the temperature (2.3 RT/ F ≃ 60 mV at T = 300 K). The IMF is defined as the sum of these two contributions by Ions move according to both electrical and concentration gradients. As a consequence, important quantities like the elementary step size and mechanisms like the force generation are difficult to resolve experimentally and model theoretically. For example, under physiological conditions, the energy provided by the translocation of a single ion is about three times smaller than that released by ATP hydrolysis. Given an energy source that is a charged quantized particle, moving along an electric field, both the experimental and theoretical treatments of the BFM energization are challenging. The demonstration that the motor is driven not by the energy of ATP hydrolysis but by the flux of ions across the membrane ( Larsen et al., 1974 Manson et al., 1977) came a few years after the realization that flagella rotate ( Berg and Anderson, 1973). Flagellar rotation is driven by the ion-motive force (IMF), the electro-chemical potential difference built across the membrane during cellular respiration. One of the few known examples of biological rotatory machines, the BFM is unique in its remarkable power and efficiency in converting free energy into mechanical work. The bacterial flagellar motor (BFM) is the powerful molecular nanomachine at the base of each flagellum responsible for such rotation. Several bacteria propel themselves by rotating their flagella ( Berg and Anderson, 1973). Perfecting our incomplete understanding of how the BFM employs the energy of ion flow will help decipher the dynamical behavior of the bacterial IMF. Within the growing toolbox of single cell bacterial electrophysiology, one of the best tools to probe IMF fluctuations may, ironically, be the motor that consumes it. Finally, while long assumed to be static and homogeneous, there is mounting evidence that the IMF is dynamic, and that its fluctuations control important phenomena such as cell-to-cell signaling and mechanotransduction. the rate of ion flow through the stators. This, in turn, raises the question of to what degree the classical dependence of motor speed on IMF is due to stator dynamics vs. We examine a series of studies that establish a linear proportionality between IMF and motor speed, and we discuss more recent evidence that the stator units sense the IMF, altering their rates of dynamic assembly. Here, we review some of the main results that form the base of our current understanding of the relationship between the IMF and the functioning of the flagellar motor. 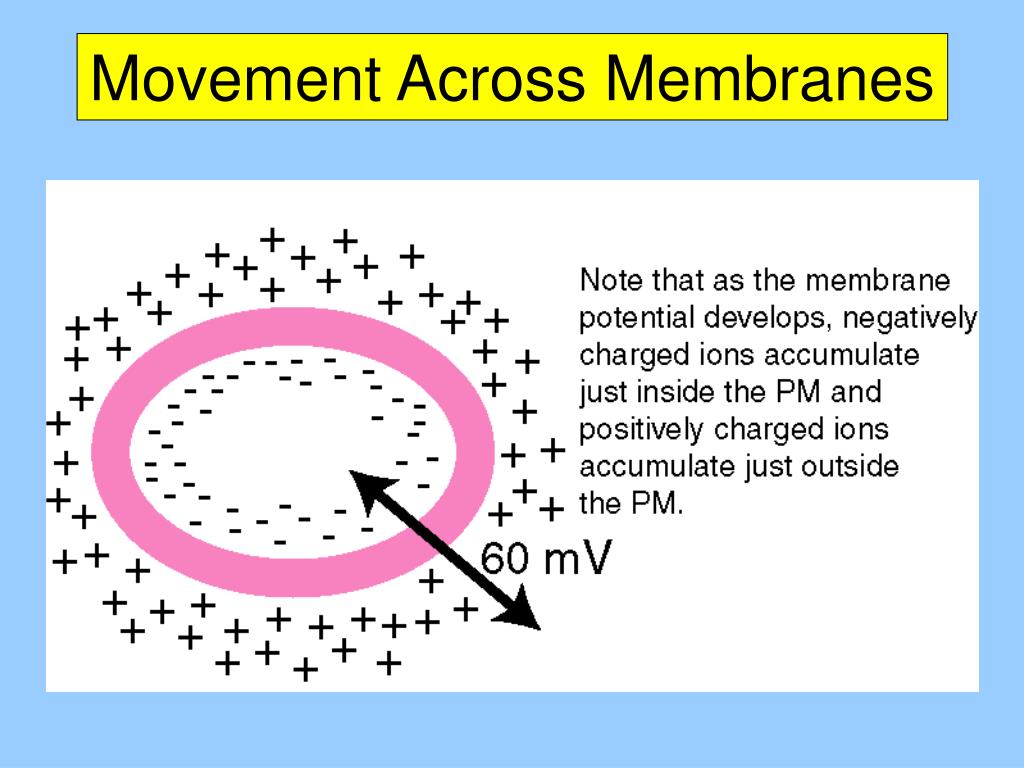 
Stator units consume the ion motive force (IMF), the electrochemical gradient across the inner membrane that results from cellular respiration, converting the electrochemical energy of translocated ions into mechanical energy, imparted to the rotor. The BFM is rotated by stator units, inner membrane protein complexes that stochastically associate to and dissociate from individual motors at a rate which depends on the mechanical and electrochemical environment. It turns a flagellum which acts as a propeller, enabling bacterial motility and chemotaxis. The bacterial flagellar motor (BFM) is a rotary molecular motor embedded in the cell membrane of numerous bacteria. Centre de Biologie Structurale (CBS), INSERM, CNRS, Université Montpellier, Montpellier, France.Anaïs Biquet-Bisquert Gilles Labesse Francesco Pedaci Ashley L.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
